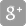Sanofi Q3 2020 business EPS(1) growth of 8.8% at CER
Q3 2020 sales growth(2) driven by strong Dupixent® and flu vaccines demand.
- Net sales were €9,479 million, up 5.7%(2) at CER and down 0.2% on a reported basis.
- Specialty Care sales grew 23.8%, driven by strong Dupixent® performance (+68.6% to €918 million) and growth in all franchises.
- Vaccines sales up 13.6%, driven by record flu sales partly offset by decline in boosters, travel and Menactra® due to COVID-19.
- General Medicines sales single-digit decline (-6.4%), reflecting China VBP program and U.S. Diabetes.
- CHC sales down 1.1% due to Zantac® voluntary recall in October 2019 and lower in-person pharmacy traffic in Rest of the World.
Q3 2020 business EPS(1) growth at CER driven mainly by strong sales performance and efficiencies.
- Q3 2020 business net income was up 1.0% on a reported basis to €2,299 million and increased 9.4% at CER.
- Q3 2020 business EPS(1) was €1.83, up 0.5% on a reported basis and 8.8% at CER.
- Q3 2020 IFRS EPS was €1.55.
Delivering on R&D
- Adding seven new Phase 3 programs to our oncology and immunology pipelines.
- COVID-19 adjuvanted recombinant protein-based vaccine Phase 1/2 trial fully enrolled.
- Principia acquisition brings full ownership of brain-penetrant BTKi tolebrutinib and innovative BTKi platform.
2020 business EPS(1) guidance revised upward
• Sanofi now expects 2020 business EPS(1) to grow between 7% and 8%(3) at CER, barring unforeseen major adverse events. Applying average October 2020 exchange rates, the currency impact on 2020 business EPS is estimated to be between -6% to -7%.
Sanofi Chief Executive Officer, Paul Hudson, commented:
“We achieved a strong quarter supported by solid sales from both Dupixent® and Vaccines which allows us to upgrade our full year guidance. We remain focused on executing on our strategic priorities that will deliver promising medicines to address significant patient needs. To this end, we strengthened our R&D pipeline with the successful completion of the Principia acquisition, adding multiple BTK inhibitors to address a variety of serious illnesses. Our COVID-19 vaccines development efforts continue on a fast track along with ensuring global access with pre-orders signed with major countries, regions, and non-profit organizations who will work to distribute the vaccine to those who most need it.”
.
| Q3 2020 | Change | Change at CER | 9M 2020 | Change | Change at CER | |
| IFRS net sales reported | €9,479m | -0.2% | +5.7% | €26,659m | +0.5% | +3.0% |
| IFRS net income reported | €1,952m | +10.5% | — | €11,233m | +298.9% | — |
| IFRS EPS reported | €1.55 | +9.9% | — | €8.96 | +298.2% | — |
| Free cash flow(4) | €1,884m | +4.7% | — | €5,452m | +39.7% | — |
| Business operating income | €3,027m | +1.0% | +9.2% | €7,710m | +5.6% | +9.6% |
| Business net income(1) | €2,299m | +1.0% | +9.4% | €5,820m | +5.5% | +9.6% |
| Business EPS(1) | €1.83 | +0.5% | +8.8% | €4.64 | +5.0% | +9.3% |
(1) In order to facilitate an understanding of operational performance, Sanofi comments on the business net income statement. Business net income is a non-GAAP financial measure (definition in Appendix 7). The consolidated income statement for Q3 2020 is provided in Appendix 3 and a reconciliation of reported IFRS net income to business net income is set forth in Appendix 4; (2) Changes in net sales are expressed at constant exchange rates (CER) unless otherwise indicated (definition in Appendix 7); (3) 2019 restated business EPS was €5.64, reflecting the discontinuation of equity method accounting for Regeneron investment; (4) Free cash flow is a non-GAAP financial measure (definition in Appendix 7).
2020 third-quarter and first nine months Sanofi sales
----------------------------
Unless otherwise indicated, all percentage changes in sales in this press release are stated at CER1
----------------------------
In the third quarter of 2020, Sanofi sales were €9,479 million, down 0.2% on a reported basis. Exchange rate movements had a negative effect of 5.9 percentage points, mainly driven by the decrease of the U.S. dollar, Brazilian real, Turkish lira, Argentine and Mexican pesos, Russian ruble and Chinese yuan. At CER, Sanofi sales increased 5.7%.
In the first nine months of 2020, Sanofi sales reached €26,659 million, up 0.5% on a reported basis. Exchange rate movements had a negative effect of 2.5 percentage points. At CER, Sanofi sales were up 3.0%.
1 See Appendix 7 for definitions of financial indicators.
Global Business Units
2020 third-quarter and first nine months business operating income
Third-quarter business operating income (BOI) increased 1.0% to €3,027 million. At CER, BOI increased 9.2%. The ratio of BOI to net sales increased 0.3 percentage points to 31.9% versus the prior year. In the first nine months, BOI increased 5.6% to €7,710 million. At CER, BOI increased 9.6%. The ratio of business operating income to net sales increased 1.4 percentage points to 28.9%.
Pharmaceuticals
Third-quarter 2020 Pharmaceutical sales increased 4.5% to €6,361 million, with double-digit growth of the Specialty Care portfolio mainly driven by the strong performance of Dupixent® which largely offset lower sales in General Medicines due to pricing pressures from the VBP (volume-based procurement) program in China and the Diabetes franchise in the U.S. First nine months Pharmaceuticals sales increased 3.3% to €19,381 million driven by the strong performance of Specialty Care.
Specialty Care
Dupixent
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Total Dupixent® | 918 | +68.6 | % | 2,552 | +83.5 | % | ||
Dupixent® (collaboration with Regeneron) generated sales of €918 million in the third quarter (up 68.6%). In the U.S., Dupixent® sales of €725 million (up 67.7%) were driven by continued strong demand in atopic dermatitis (AD) in adult and adolescent patients, rapid adoption in children aged 6 to 11 years (approved in May 2020) and continued uptake in asthma. Dupixent® total prescriptions (TRx) increased 80% (year-over-year) and new-to-brand prescriptions (NBRx) grew 25% despite fewer in-person physician visits which remain below the pre-COVID level. In Europe, third-quarter Dupixent® sales grew 73.2% to €97 million reflecting continued growth in AD in key countries and additional launches in asthma in other European markets. In Japan, sales were €48 million (up 31.6%), where strong demand was moderated by the government price decrease implemented in April 2020. Dupixent® was approved in China for the treatment of adults with moderate-to-severe AD in June and more than 1,100 patients have already been treated. First nine months Dupixent® sales reached €2,552 million, up 83.5% reflecting increased penetration into eligible AD and asthma populations as well as expansion in additional geographies and new indications in younger populations.
Multiple Sclerosis/Neurology/Other Inflammation & Immunology
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Aubagio® | 505 | +6.7 | % | 1,573 | +13.0 | % | ||
| Lemtrada® | 24 | -56.1 | % | 92 | -58.3 | % | ||
| Kevzara® | 59 | +28.6 | % | 176 | +35.9 | % | ||
| Total Multiple Sclerosis/ Neurology/Other I&I | 588 | +2.5 | % | 1,841 | +5.7 | % | ||
Third-quarter and first nine months Multiple Sclerosis/Neurology/Other I&I sales increased 2.5% (to €588 million) and 5.7%, respectively driven by Aubagio® and Kevzara® sales growth.
Aubagio® sales increased 6.7% in the third quarter to €505 million, driven by Europe (up 15.4%), mainly benefiting from demand growth and a price increase in Germany. In the U.S., Aubagio® sales grew 2.8% reflecting continued strong adherence and pricing benefit, which offset the unwinding of COVID-19 related patient stocking and lower new patient starts. In the first nine months, Aubagio® sales increased 13.0% mainly driven by stronger demand and price increases in the U.S and Germany.
Third-quarter and first nine months Lemtrada® sales decreased 56.1% (to €24 million) and 58.3%, respectively, primarily due to the COVID-19 pandemic, which has led to a decrease in infused immune reconstitution therapies such as Lemtrada®.
Third-quarter and first nine months Kevzara® (collaboration with Regeneron) sales were up 28.6% (to €59 million) and 35.9%, respectively, reflecting launch dynamics and modest COVID-19 impact.
Rare Disease
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Myozyme® / Lumizyme® | 241 | +11.5 | % | 713 | +6.8 | % | ||
| Fabrazyme® | 204 | +6.4 | % | 617 | +4.7 | % | ||
| Cerezyme® | 162 | +6.0 | % | 530 | +5.8 | % | ||
| Aldurazyme® | 55 | +20.4 | % | 177 | +7.1 | % | ||
| Cerdelga® | 60 | +18.9 | % | 175 | +17.2 | % | ||
| Others Rare Disease | 23 | +4.3% | 65 | +4.8% | ||||
| Total Rare Disease | 745 | +9.7 | % | 2,277 | +6.7 | % | ||
In the third quarter, Rare Disease sales increased 9.7% to €745 million, reflecting favorable order phasing in Rest of the World and partial COVID-19 recovery largely in the U.S. First nine months Rare Disease sales increased 6.7% driven by Rest of the World.
Third-quarter Cerezyme® sales increased 6.0% to €162 million, primarily due to favorable order phasing in the Rest of the World. Third-quarter Cerdelga® sales increased 18.9% to €60 million driven by patient accruals largely in the U.S. (up 20.0%). Sales of the Gaucher franchise (Cerezyme® + Cerdelga®) increased 9.0% (to €222 million) in the third quarter and 8.4% in the first nine months mainly driven by favorable order phasing and new patient accruals in the Rest of the World.
Third-quarter Myozyme®/Lumizyme® sales increased 11.5% to €241 million supported by new patient accruals, patients returning to treatment, and inventory build in the U.S. as well as favorable order phasing in Rest of the World. First nine months Myozyme®/Lumizyme® sales increased 6.8% driven by new patient accruals in the U.S. and Rest of the World.
Third-quarter Fabrazyme® sales increased 6.4% to €204 million reflecting patients returning to treatment in the U.S. and sales growth in the Rest of the World despite competition and price reduction in Japan. Fabrazyme® was launched in China in May and is the first treatment for Fabry disease to be approved in China. First nine months Fabrazyme® sales were up 4.7%, reflecting new patient accruals in the U.S. and Europe, partially offset by COVID-19 impact.
Oncology
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Jevtana® | 134 | +16.0 | % | 405 | +14.0 | % | ||
| Fasturtec® | 42 | +29.4 | % | 114 | +16.2 | % | ||
| Libtayo® | 21 | +425.0 | % | 48 | — | |||
| Sarclisa® | 13 | — | 18 | — | ||||
| Total Oncology | 210 | +37.6 | % | 585 | +28.1 | % | ||
Third-quarter Oncology sales increased 37.6% to €210 million, driven by Sarclisa® and Libtayo® launches and growth from legacy franchises. In the first nine month, Oncology sales were up 28.1% supported by growth in all three regions.
Third-quarter Jevtana® sales increased 16.0% to €134 million driven by strong demand in the U.S. (up 34.0%) and Europe (up 14.3%). Sales performance benefited from increased demand in metastatic castration-resistant prostate cancer following publication of the results of the CARD study in this disease setting at ESMO (European Society for Medical Oncology) and in the NEJM (New England Journal of Medicine) in September 2019. First nine months sales of Jevtana® were up 14.0% driven by the U.S. performance. In the U.S., the Jevtana® composition of matter patent will expire in September 2021. From May to July 2020, Sanofi filed patent infringement suits against all generic filers on Jevtana® under Hatch-Waxman in the U.S. District Court for the District of Delaware asserting two method of use patents (US 10,583,110 and US 10, 716,777), both of which lasts until October 2030.
Libtayo® (collaboration with Regeneron) approved for the treatment of patients with metastatic cutaneous squamous cell carcinoma (CSCC) or locally advanced CSCC who are not candidates for curative surgery or curative radiation had ex-U.S. sales of €21 million in the third quarter and €48 million in the first nine months. Sales were driven by new country launches. To date, Libtayo® has been launched in 17 countries outside the U.S. and up to 7 additional country launches are planned by the end of 2020. U.S. Libtayo® sales are reported by Regeneron.
Sarclisa® was approved in March in the U.S. for the treatment of adults with relapsed refractory multiple myeloma (RRMM) who have received at least two prior therapies including lenalidomide and a proteasome inhibitor and in June by the European Commission in certain adults with RRMM. Third-quarter Sarclisa® sales were €13 million of which €9 million was generated in the U.S. despite the challenging launch environment due to COVID-19. Sarclisa® is now launched in the U.S., Austria, Japan, Switzerland, Canada and UK. Five additional commercial launches are expected by year-end.
Rare Blood Disorder
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Eloctate® | 152 | -1.9 | % | 482 | -5.3 | % | ||
| Alprolix® | 109 | +10.6 | % | 335 | +10.5 | % | ||
| Cablivi® | 31 | +65.0 | % | 83 | +110.0 | % | ||
| Total Rare Blood Disorder | 292 | +7.3 | % | 900 | +5.8 | % | ||
In the third quarter, Rare Blood Disorder franchise sales were €292 million, up 7.3% driven by Cablivi® and Alprolix® performance. First nine months franchise sales increased 5.8% driven by these two products which largely offset Eloctate® sales decline in the U.S.
Eloctate® sales were €152 million in the third quarter, down 1.9% due to lower U.S. sales (-6.6%) as a result of ongoing competitive pressure. In the Rest of the World, Eloctate® sales increased 12.5% driven by phasing of sales following changes to the supply relationship with Sobi more than offsetting lower sales in Japan due to a price reduction and competitive pressures. Eloctate® sales to Sobi are expected to be volatile for the foreseeable future. First nine months Eloctate® sales decreased 5.3% driven by competitive pressure in the U.S.
Alprolix® sales were €109 million in the third quarter, up 10.6%, mainly driven by patient switches from short-acting factors, prophylaxis conversion and increased sales to Sobi (Rest of the World sales up 24.0%) following changes to the supply relationship with Sobi. Alprolix® sales to Sobi are expected to be volatile for the foreseeable future. First nine months Alprolix® sales increased 10.5% as a result of the U.S performance and sales to Sobi.
Cablivi® for the treatment of adults with acquired thrombotic thrombocytopenic purpura (aTTP) generated third-quarter sales of €31 million mainly driven by the U.S., Germany and successful launch in Italy. In Europe, the product is commercially available in several countries and has a temporary license to be sold in France. First nine months Cablivi® sales were €83 million reflecting sales in the U.S. as well as additional launches in Europe. In July; the International Society on Thrombosis and Haemostasis (ISTH) published the first evidence based global guidelines on the diagnosis and treatment of thrombotic thrombocytopenic purpura (TTP) in the Journal of Thrombosis and Haemostasis. The ISTH guidelines suggest treatment with Cablivi® in combination with plasma exchange and immunosuppressive therapy for first episode or relapse in all adult aTTP patients.
General Medicines
Diabetes
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Lantus® | 657 | -7.1 | % | 2,074 | -6.9 | % | ||
| Toujeo® | 216 | +3.7 | % | 712 | +11.4 | % | ||
| Total glargine | 873 | -4.6 | % | 2,786 | -2.8 | % | ||
| Apidra® | 79 | +2.4 | % | 252 | +2.7 | % | ||
| Admelog® | 48 | -2.0 | % | 142 | -26.8 | % | ||
| Soliqua® | 40 | +27.3 | % | 115 | +41.0 | % | ||
| Other diabetes | 106 | -12.8 | % | 327 | -12.1 | % | ||
| Total Diabetes | 1,146 | -4.0 | % | 3,622 | -3.6 | % | ||
In the third quarter, global Diabetes sales decreased 4.0% to €1,146 million, due to a continued decline in the average U.S. glargine price (Lantus® and Toujeo®), lower Amaryl® sales in China as well as some COVID-19 impact, mainly in the Rest of the World. First nine months franchise sales were down 3.6% mainly due to lower glargine sales in the U.S.
Lantus® sales were €657 million in the third quarter, down 7.1%. mainly due to a continued decline in average U.S. price, patients switching to Toujeo® and biosimilar glargine competition. In Rest of the World, sales decreased 2.9%, reflecting some COVID-19 impact on the out-of-pocket market as well as an unfavorable phasing effect. First nine months Lantus® sales decreased 6.9%, mainly due to lower sales in the U.S. and to a lesser extent in Europe.
Third-quarter Toujeo® sales increased 3.7% to €216 million, driven by Rest of the World and Europe, which benefited from switches from Lantus® and new patient starts. In the U.S., third-quarter Toujeo® sales decreased 4.1% due to a continued decline in the average U.S. price which more than offset volume growth. Toujeo was approved in China in the third quarter and will be launched at the end of the year. First nine months Toujeo® sales increased 11.4%, driven by strong performance in Rest of the World and Europe.
Amaryl® sales decreased 16.7% in the third quarter to €67 million, due to lower sales in China (-44.4% to €20 million) reflecting the second wave of the VBP program which includes glimepiride (compound name of Amaryl®). As previously disclosed, Sanofi opted not to participate in the bidding for Amaryl® and expects sales in China for the brand to decline significantly in 2020. First nine months Amaryl® sales decreased 18.4% to €204 million reflecting the impact from VBP in China from April.
Third-quarter Soliqua®/Suliqua® sales increased 27.3% to €40 million driven by launches in Rest of the World. In the first nine months, Soliqua® sales increased 41.0% supported by strong growth in all three regions.
Cardiovascular and Established Rx Products
| Net sales (€ million) | Q3 2020 | Change at CER | 9M 2020 | Change at CER | ||||
| Lovenox® | 365 | +17.1 | % | 995 | +1.5 | % | ||
| Plavix® | 205 | -39.9 | % | 714 | -35.6 | % | ||
| Aprovel®/Avapro® | 133 | -17.8 | % | 439 | -17.5 | % | ||
| Thymoglobulin® | 87 | +1.1 | % | 236 | -9.4 | % | ||
| Multaq® | 79 | -5.7 | % | 233 | -6.0 | % | ||
| Praluent® | 50 | -16.4 | % | 196 | +6.6 | % | ||
| Renvela®/Renagel® | 63 | -22.4 | % | 194 | -14.8 | % | ||
| Synvisc® /Synvisc-One® | 56 | -19.2 | % | 152 | -32.0 | % | ||
| Mozobil® | 56 | +16.0 | % | 155 | +9.1 | % | ||
| Eloxatin® | 52 | +1.9 | % | 146 | -7.5 | % | ||
| Taxotere® | 41 | +2.4 | % | 119 | -7.6 | % | ||
| Generics | 219 | -3.4 | % | 713 | -2.2 | % | ||
| Other | 1,056 | -4.4 | % | 3,312 | -4.9 | % | ||
| Total Cardiovascular and Established Rx Products | 2,462 | -7.5 | % | 7,604 | -9.4 | % | ||
In the third quarter, Cardiovascular and Established Rx Products sales decreased 7.5% to €2,462 million, primarily driven by the decline in Plavix® and Aprovel® family sales in China, some COVID-19 impact, mainly in the Rest of the World.
Third-quarter Lovenox® sales increased 17.1% to €365 million, driven by Rest of the World (up 36.5% to €189 million), reflecting recent guidelines recommending the use of low molecular weight heparins in hospitalized COVID-19 patients. In Europe, Lovenox® sales were up 0.6% to €169 million benefiting from the catch-up of elective procedures which has offset biosimilar competition in several countries. First nine months Lovenox® sales were up 1.5% driven by Rest of the World sales growth which offset biosimilar competition in Europe.
Plavix® sales were down 39.9% in the third quarter to €205 million, primarily reflecting the decrease in China (sales down 64.1% to €72 million) due to net price adjustments following the implementation of the VBP program partially offset by volume gains. In Japan, Plavix® sales decreased 25.0% to €24 million due to a price reduction in October 2019. First nine months Plavix® sales decreased 35.6%, reflecting lower sales in China.
Third-quarter Aprovel®/Avapro® sales were down 17.8% to €133 million, primarily reflecting the decrease in China (sales down 37.8% to €44 million) due to lower net price following the implementation of the VBP program partially offset by volume gains. First nine months Aprovel®/Avapro® sales decreased 17.5%, reflecting lower sales in China.
As previously announced, Sanofi expects sales of Plavix® and the Aprovel® family in China to decline by around 50% in 2020 due to the implementation of the VBP program. In the third quarter, volume growth of Plavix® and CoAprovel® increased more than 60% in China in line with Sanofi’s full-year expectations.
Third-quarter Praluent® sales decreased 16.4% to €50 million, due to lower sales in the U.S. which more than offset performance in Europe (up 25.0% to €30 million). Praluent® was launched in China in April. Effective April 1, 2020, Sanofi has sole responsibility for Praluent® outside the U.S. while Regeneron has sole responsibility for Praluent® in the U.S. Both companies have entered into agreements to support manufacturing needs in the near term and Sanofi will book sales of Praluent® in 2020 in the U.S. First nine months Praluent® sales grew 6.6% driven by the U.S. and Rest of the World.
Pharmaceuticals business operating income
In the th

Upcoming Life Sciences Events
- April 2024
- Singapore: Asia Bio Partnering Forum
- London: LSX World Congress
- May 2024
- Seoul: BIO KOREA 2024
 Biotechgate Global Database ›
Biotechgate Global Database ›